Another Epigenetics co get bought out, not many left
posted on
Jul 10, 2022 08:38AM

Zenith's BET Inhibitor ZEN-3694 is Currently Being Evaluated in Multiple Oncology Clinical Trials

Well a buyout offer is an offer, Compared to Constellation Pharma this one valuation is far less but the long term revenue on their Oncology drug was limited and not large. Zenith Epi IMO has far far more long term potential and could fetch for ten times more similar to Constellation valauation. It just goes to show you Pharma is indeed interest in this field. Btw Epizyme traded at $20usd a year or so ago .
Shares of Epizyme EPZM surged 60% in pre-market trading on Jun 27 after management announced that it has signed a definitive merger agreement with Europe-based Ipsen IPSEY, wherein the latter will acquire all the outstanding shares for an estimated aggregate consideration of $247 million.
Per the agreement terms, Ipsen will pay upfront cash of $1.45 per share to current Epizyme shareholders, representing a premium of 52% to EPZM’s closing price on Jun 24.
In addition, the existing shareholders will also be eligible to receive one contingent value right (“CVR”) per share. A CVR of $0.30 per share will be paid by IPSEY if, by Dec 31, 2026, the aggregate sales of Tazverik (excluding sales in Japan and Greater China) in any four consecutive quarters cross the $250 million threshold. A further $0.70 per share will also be payable by Ipsen if the combination of Tazverik with R2 (rituximab and lenalidomide) receives FDA approval for use in second-line follicular lymphoma by Jan 1, 2028.
Shares of Epizyme have plunged 61.8% in the year so far in comparison with the industry’s 21.7% decline.
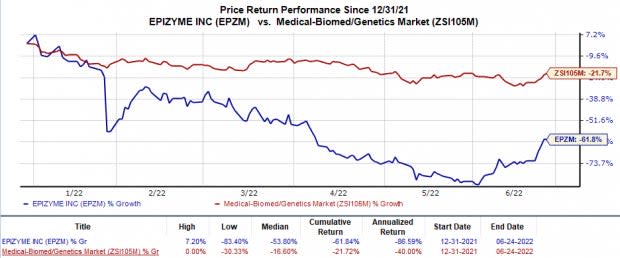
Image Source: Zacks Investment Research
Currently, Epizyme has only one marketed drug in its portfolio, Tazverik (tazemetostat). A methyltransferase inhibitor, Tazverik was approved by the FDA in January 2020, to treat adults and pediatric patients aged 16 years and above with metastatic or locally advanced epithelioid sarcoma (ES) not eligible for complete resection. The drug also approved the label expansion for Tazverik in June 2020 to treat two distinct follicular lymphoma (FL) indications in adult patients. Both indications are approved by the FDA under the accelerated pathway.
As part of the accelerated approval for ES & FL, the company is evaluating Tazverik in separate ES & FL confirmatory studies. The ES confirmatory study is evaluating Tazverik in combination with doxorubicin compared with doxorubicin plus placebo as a front-line treatment for ES. Additionally, the FL confirmatory study is evaluating Tazverik in combination with R2, compared with R2 plus placebo for treating relapsed/refractory FL patients.